2026.04.08 Wednesday
Antibody Drugs with Strong Immune Cell Binding Linked to Allergic Reactions
Chiba University
Tokyo University of Science
Study finds that stronger Fcγ receptor binding associated with increased anaphylaxis risk in preclinical models
Antibody therapies have been used to treat diseases for nearly two decades, but their association with harmful immune reactions, including anaphylaxis, remains unclear. Researchers from Chiba University examined how antibodies interact with Fcγ receptors in tumor-bearing mouse models and found that strong Fcγ receptor binding was associated with higher antidrug antibody levels and anaphylaxis, with tumor-associated myeloid cells implicated in this process. These findings may help guide the development of safer antibody therapies.
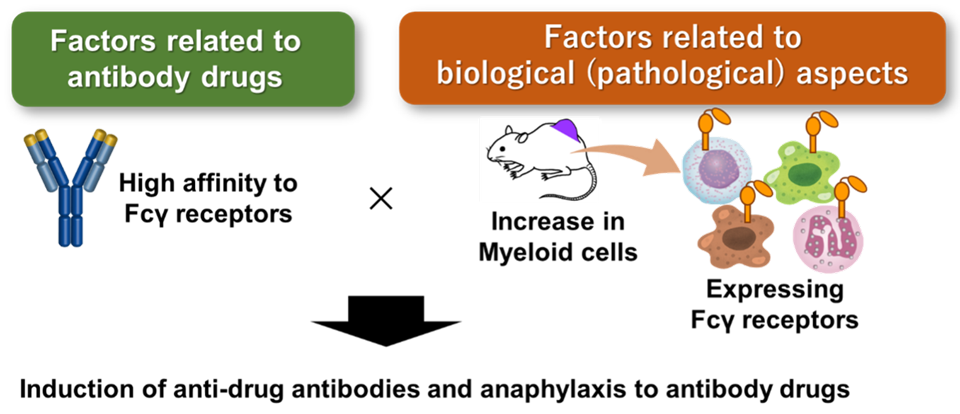
Image title: Fcγ Receptor Binding is Linked to Antibody Anaphylaxis
Image caption: In tumor-bearing mouse models, anti-programmed death-ligand 1 antibodies with stronger Fcγ receptor binding were associated with higher antidrug antibody levels and anaphylaxis, with tumor-associated myeloid cells implicated in the capture of these antibodies.
Image credit: Professor Hiroto Hatakeyama from Chiba University, Japan
Image source link: N/A
Image license: Original content
Usage Restrictions: Cannot be reused without permission.
Antibody therapeutics are laboratory-made proteins designed to work like the body's natural antibodies. They are widely used to treat diseases such as cancer by binding to specific targets, including cancer cells or inflammatory molecules. However, in some cases, the body may recognize these drugs as foreign and produce antidrug antibodies (ADAs) to eliminate them.
This immune response, in some cases, can lead to anaphylaxis, a severe and potentially life-threatening allergic reaction. Although rare, such reactions can occur suddenly and are difficult to predict, making them a major safety concern in antibody-based therapies. Despite this risk, the exact reasons why some antibody drugs trigger ADAs and anaphylaxis are not fully understood.
To investigate this, researchers from Chiba University, Japan, examined how antibody therapeutics interact with immune receptors called Fcγ receptors, which are found on certain immune cells and help recognize antibodies. The study was conducted by Professor Hiroto Hatakeyama and Dr. Ruiheng Tang from the Graduate School of Pharmaceutical Sciences, Chiba University, along with Dr. Kyohei Higashi from the Faculty of Pharmaceutical Sciences, Tokyo University of Science, Japan.
Their findings, published in Volume 14, Issue 3 of the Journal for ImmunoTherapy of Cancer on March 4, 2026, suggest that drugs with stronger binding to Fcγ receptors are more likely to be recognized as foreign by the immune system and are associated with higher ADA production.
"Our focus is on fatal anaphylaxis resulting from excessive ADA production. We find that high affinities for Fcγ receptors are critical determinants of anaphylaxis. Our findings reveal a mechanism linking Fcγ receptor-mediated antibody capture by tumor-associated myeloid cells to ADA induction," says Prof. Hatakeyama.
Anaphylaxis has traditionally been explained by the immunoglobulin E (IgE) pathway. In this pathway, exposure to an antigen activates B lymphocytes, which then produce IgE antibodies. These antibodies bind to receptors on mast cells and basophils, causing them to release substances such as histamine, which leads to allergic symptoms. However, growing evidence suggests that anaphylaxis can also occur through IgE-independent pathways.
To explore this, the researchers conducted experiments in tumor-bearing mice. They tested two antibodies that both target programmed death-ligand 1 (PD-L1), a protein found on cancer cells that helps them evade the immune system. When 10F.9G2, an antibody with a strong ability to bind Fcγ receptors on immune cells, was administered, all mice rapidly developed fatal anaphylaxis, accompanied by a sharp increase in ADA levels. In contrast, another PD-L1-targeting antibody, MIH6, which has a lower binding affinity for Fcγ receptors, was not associated with this reaction and showed very low levels of ADAs.
To confirm the role of this interaction, the researchers created modified versions of 10F.9G2 with reduced Fcγ receptor binding. These modified antibodies did not trigger anaphylaxis and were associated with low ADA production, supporting the idea that strong Fcγ receptor interactions drive the response.
The study also suggests that tumor-associated myeloid cells may play an important role. These cells were found to capture antibodies with strong Fcγ receptor binding and process them in a way that may promote immune activation, which was associated with increased ADA production.
Importantly, when Fcγ receptors were blocked, this process was greatly reduced. The immune cells showed limited ability to capture antibodies, and this was associated with lower ADA levels and improved survival in the mice. This observation suggests that Fcγ receptor interactions may be a potential target for reducing the risk of anaphylaxis.
The researchers also looked at clinical data from the Food and Drug Administration Adverse Events Reporting System database and found a similar pattern: antibody drugs with stronger Fcγ receptor binding or higher antibody-dependent cellular cytotoxicity activity, which is a measure of immune cell activation, were more frequently associated with anaphylaxis.
Overall, the study provides insight into how antibody therapeutics may be associated with anaphylaxis. It highlights a possible link between Fcγ receptor binding, ADA production, and immune reactions, and may help guide the development of safer antibody therapies in the future.
"Since this research is based on a tumor model, it is unlikely to have immediate effects on daily life. However, we hope it will help explain how antibody drugs cause anaphylaxis in clinical settings," says Prof. Hatakeyama.
Reference
| Title of original paper | : | Antibody therapeutics with high affinity for FcγRs exacerbate anaphylaxis via FcγR-mediated capture by tumor-associated myeloid cells |
| Authors | : | Ruiheng Tang1, Aizemaiti Aibai1, Yuta Tamemoto1, Riho Kume1, Kazuto Yasuda1, Kenta Sato1, Hirohito Abo2, Kazuto Tsuji3, Kyohei Higashi3, Masato Kiyoshi4, Noritaka Hashii4, Akiko Ishii-Watabe4, Hiroto Kawashima5, and Hiroto Hatakeyama1,6 |
| Affiliations | : | 1Laboratory of DDS Design and Drug Disposition, Graduate School of
Pharmaceutical Sciences, Chiba University, Japan 2Center for Inflammation, Immunity and Infection, Institute for Biomedical Sciences, Georgia State University, United States 3Faculty of Pharmaceutical Sciences, Tokyo University of Science, Japan 4Division of Biological Chemistry and Biologicals, National Institute of Health Sciences, Japan 5Laboratory of Microbiology and Immunology, Graduate School of Pharmaceutical Sciences, Chiba University, Japan 6Center of Quantum Life Science for Structural Therapeutics, Chiba University, Japan |
| Journal | : | Journal for ImmunoTherapy of Cancer |
| DOI | : | 10.1136/jitc-2025-013316 |
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society," TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
■
Tokyo University of Science(About TUS)

About Associate Professor Kyohei HIGASHI
from Tokyo University of Science
Funding
The study was funded by JSPS KAKENHI of Transformative Research Areas (A) "Material Symbiosis" (grant number: 21H05506); the Early-Stage Research Grant for Medical and Pharmaceutical Sciences from the Future Medicine Education and Research Organization at Chiba University; the Waksman Foundation of Japan; Hoyu Science Foundation; TELUMO Life Science Foundation; JST SPRING (grant number: JPMJSP2109); cSIMVA Vaccine Challenge Grants; and the Japan Agency for Medical Research and Development (grant number: JP21ae0121013).

