2026.03.19 Thursday
How Inflammation Drives Bone Loss in an Aggressive Childhood Leukemia
Researchers identify an inflammatory feedback loop that accelerates disease progression and damages bones, pointing to new therapeutic targets
A rare form of leukemia known as TCF3::HLF-positive B cell acute lymphoblastic leukemia (B-ALL) sits among the most aggressive blood cancers seen in children. The disease causes a rapid buildup of abnormal blood cells, but unlike other types of leukemia, it also severely damages bones and causes pain. To make things worse, patients with TCF3::HLF-positive B-ALL tend to relapse quickly after treatment, with survival rates remaining low.
Despite decades of research, progress against this aggressive leukemia has been slow and unremarkable. One major obstacle has been the lack of animal models that accurately reflect the disease as seen in humans, making it difficult to study how the disease develops. At the same time, while bone loss and inflammation have been frequently observed together in patients, scientists cannot explain why leukemia cells trigger these effects nor tell if they are connected to the cancer's rapid growth and resistance to treatment.
Fortunately, in a study that was published online in the journal Blood on March 03, 2026, a research team led by Professor Tomokatsu Ikawa from the Research Institute for Biomedical Sciences, Tokyo University of Science (TUS), Japan, took a major step in addressing these challenges. Their paper, co-authored by a third-year doctoral student, Ms. Aisa Suzuki from TUS and Dr. Takeshi Inukai from the University of Yamanashi, Japan, describes the development of a new laboratory mouse model that closely mirrors TCF3::HLF-positive B-ALL observed in humans. Using this model, the researchers uncovered a previously unknown mechanism that links inflammation, leukemia progression, and bone destruction.
To build their animal model, the team used specially engineered precursor blood cells. When these cells were programmed to express the TCF3::HLF fusion gene--a genetic abnormality that defines this leukemia subtype--mice developed an aggressive form of B-ALL. Through detailed observations of blood cell changes and bone damage, the researchers confirmed that the disease in mice closely matched what doctors see in patients.
Then, by analyzing gene activity in the leukemia cells, the researchers identified unusually high levels of interleukin-1 beta (IL-1β), a powerful inflammatory protein normally involved in immune responses. Unlike what is typically seen in other forms of leukemia, the leukemia cells themselves were abnormally producing and releasing IL-1β. Further experiments showed that this molecule acted like a growth signal, helping leukemia cells multiply faster while simultaneously activating bone-destroying cells called osteoclasts through a secondary signaling protein called RANKL.
Interestingly, the study also revealed how this process begins at the genetic level. The TCF3::HLF fusion protein--the hallmark of this form of leukemia--functions as an abnormal transcription factor, meaning it can switch specific genes on or off. The team discovered that it directly activates IL-1β through a previously unknown regulatory region in the genome. This mechanism appears to be specific to this leukemia subtype, which helps explain why inflammation is so pronounced in these patients. "Our findings delineate a self-reinforcing loop in which TCF3::HLF directly induces IL-1β transcription, thereby driving both leukemic expansion and bone destruction," says Prof. Ikawa. "This is an extremely remarkable discovery that not only contributes to basic research but also has potential clinical applications."
When the researchers blocked IL-1β action in diseased mice using antibodies, leukemia growth slowed, bone damage was reduced, and survival was significantly extended. The effects were even stronger when IL-1β inhibition was combined with a drug already used to treat leukemia, pointing toward new cancer treatments that could be tested in the future. "This work highlights inflammatory signaling as a promising therapeutic target in this otherwise incurable leukemia subtype," remarks Prof. Ikawa. Also, IL-1β is actively expressed in macrophages or monocytes of patients with acute myeloid leukemia, myelodysplastic syndrome, and multiple myeloma, suggesting that IL-1β could be potential target in treating these diseases.
Together, these findings provide a clearer picture of why TCF3::HLF-positive B-ALL is so aggressive and destructive. While more research is needed before these findings can be translated to the clinic, the study provides a strong foundation for testing drugs that disrupt the abnormal inflammatory signals and bone-damaging pathways. This work is expected to move to early-phase clinical evaluation within the next couple of years. Ultimately, this work moves the field closer to treatments that not only keep this cancer under control but also protect bone health and quality of life for young patients.
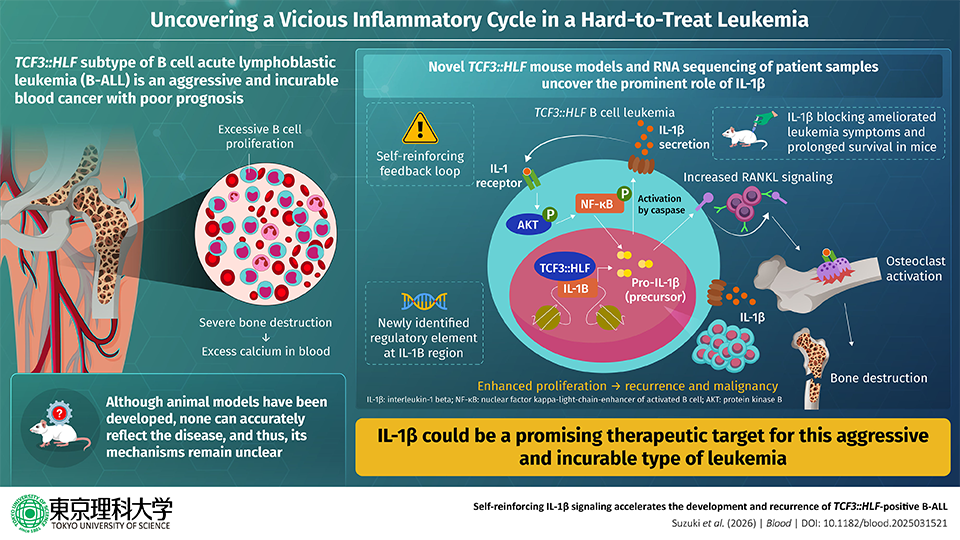
Image title: Overview of the inflammatory feedback mechanism discovered in TCF3::HLF-positive B cell acute lymphoblastic leukemia
Image caption: As depicted in the image, a newly found regulatory region in the genome leads to increased IL-1β production, leading to a self-sustaining loop. This excess IL-1β also drives the activation of osteoclasts, which ultimately causes bone damage.
Image credit: Professor Tomokatsu Ikawa from Tokyo University of Science, Japan
License type: Original content
Usage restrictions: Cannot be used without permission.
Reference
| Title of original paper | : | Self-reinforcing IL-1β signaling accelerates the development and recurrence of TCF3::HLF-positive B-ALL |
| Journal | : | Blood |
| DOI | : | 10.1182/blood.2025031521  |
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society," TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
■
Tokyo University of Science(About TUS)

About Associate Professor
Tomokatsu Ikawa
from Tokyo University of Science
Dr. Tomokatsu Ikawa is currently a Professor at the Division of Immunology and Allergy, Research Institute for Biomedical Sciences at Tokyo University of Science. He obtained a Ph.D. from Kyoto University, Japan. His work centers around hematology and immunology, including T cell development, B cell development, hematopoietic stem cells, and epigenetics. He has published more than 100 peer-reviewed papers on these topics. He is affiliated with academic societies including the Japanese Society for Interferon & Cytokine Research.
Funding information
This work was supported in part by grants from the Japan Agency for Medical Research and development; Astellas Foundation for Research on Metabolic Disorders; The Uehara Memorial Foundation; Grants for Japanese Society of Hematology; Princess Takamatsu Cancer Research Fund; The NOVARTIS Foundation (Japan) for the Promotion of Science; Foundation for Promotion of Cancer Research; Extramural Collaborative Research Grant of Cancer Research Institute, Kanazawa University; JST FOREST Program (JPMJFR205Z), and JSI Kibou Project.

