2024.07.01 Monday
New Insights into Antihistamine Binding Could Lead to More Effective Treatments
Researchers investigate why geometric variants of a small molecule drug have markedly different efficacies

Even if two molecules have the exact same chemical formula and the same number and types of bonds, their three-dimensional arrangements can still be different. While some people might mistakenly disregard this as a minor detail, even simple changes in the position or orientation of a functional group can dramatically affect the biological properties of a molecule, sometimes rendering an otherwise benign substance into a highly toxic one. Thus, the study of such possible molecular variants, called 'geometric isomers,' is essential in the field of drug development.
Doxepin stands out as a notable example of a drug that is commercialized as a mixture of two geometric isomers, namely the E- and Z-isomers. Both doxepin isomers bind to histamine H1 receptor (H1R), which is expressed throughout the central nervous system, smooth muscle cells, and vascular endothelial cells. Besides its use as an antihistaminic drug, doxepin is also typically used as an antidepressant and sleeping aid. While biological tests in animals have shown that the Z-isomer is more effective than the E-isomer, the differences in affinity to H1R between the E- and Z-isomers are unknown. Moreover, the specifics of how these compounds actually bind to H1R remain elusive.
Against this backdrop, a research team from Tokyo University of Science, Japan, set out to clarify the finer details of the interactions between doxepin isomers and H1R. Their latest paper, which was published on June 25, 2024, in the Journal of Molecular Recognition, was co-authored by Professor Mitsunori Shiroishi, Mr. Hiroto Kaneko, and Associate Professor Tadashi Ando, among others. This study is a follow-up to past work done by Prof. Shiroishi and colleagues. "We previously revealed the crystal structure of the complex formed by H1R and doxepin, but we were unable to determine which isomer was bound," he explains, "We then came up with a method to determine the binding affinity of the isomers, and thus carried out this study."
To achieve this challenging goal, the researchers first produced a customized yeast expression vector by strategically inserting the H1R gene into it. This vector was used to modify yeast cultures so that they produce H1R. After retrieving the membranes from these cells, they applied a solution containing commercial doxepin, producing H1R-doxepin complexes. Following extraction and purification of these complexes, they removed any excess (unbound) doxepin. Finally, by denaturing the H1R receptors, they could free the bound doxepin molecules and measure their numbers in a high-performance liquid chromatography setup.
Using this protocol, the researchers could accurately quantify the amount of each isomer that was bound to the extracted receptors, which is directly tied to their relative binding affinity. They found that the affinity to H1R of the Z-isomer was over five times higher than that of the E-isomer.
The team then delved deeper into the nature of how doxepin isomers bind to H1R. Through experiments on a mutant variant of H1R coupled with molecular dynamics simulations, they revealed that the Thr112 side chain in the ligand-binding pocket of H1R creates a chemical environment that enhances selectivity for the Z-isomer.
Taken together, the findings of this study shed light on how a widely used small molecule drug interacts with an important cellular receptor. "Our efforts could serve as the basis for designing next-generation antihistamines that are more effective and have fewer side effects," highlights Prof. Shiroishi, "Worth noting, this newfound knowledge will be useful for designing compounds that bind not only to H1R, but also other disease-relevant target proteins."
The rational design of future drugs, aided and validated by computational techniques like molecular dynamics simulations, could usher in a new era in medicine. More specifically, by understanding the binding properties of isomers in detail, many small-molecule drugs could be made more effective, safer, and better suited for targeted therapies.
Let us hope this vision of the future becomes a reality soon!
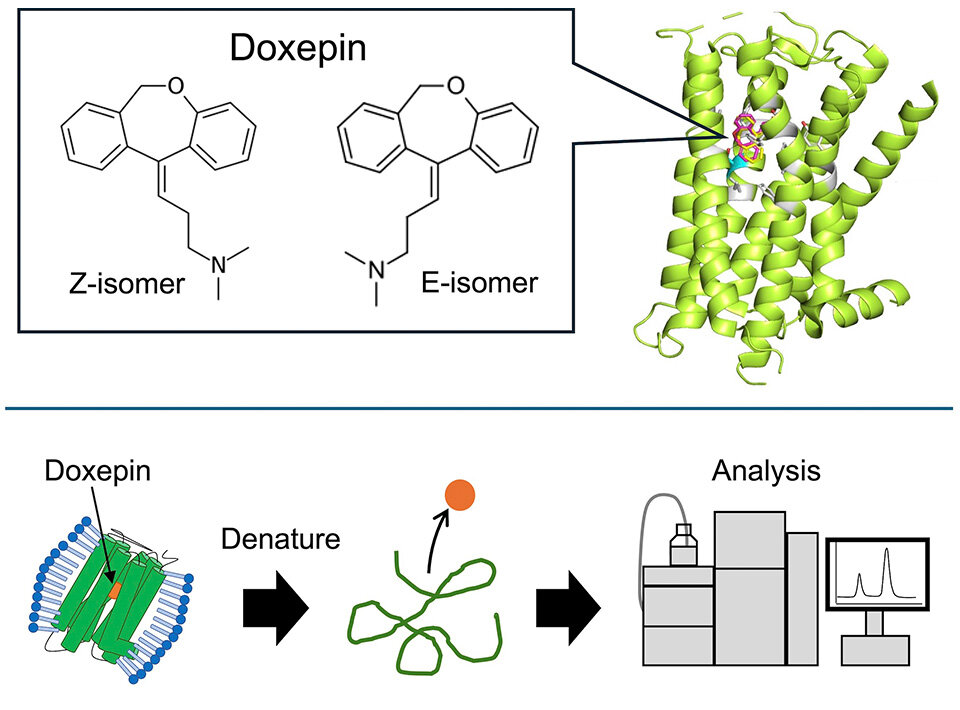
Image title: Experimental setup to determine the binding affinity of doxepin isomers to H1R
Image caption: Doxepin is an antihistaminic drug that has two geometric isomers, which bind to the H1R receptor (depicted in the top half of the image). To understand why the Z-isomer is more effective than the E-isomer, researchers developed an experimental protocol to accurately quantify the binding affinity of each isomer to H1R. This involved extracting intact H1R-doxepin complexes from yeast cultures, releasing the bound doxepin molecules, and determining which isomer they represent using high-performance liquid chromatography.
Image credit: Mitsunori Shiroishi from Tokyo University of Science
License type: Original content
Usage restrictions: You are free to share and adapt the material. Attribution is required.
Reference
| Title of original paper | : | Binding characteristics of the doxepin E/Z isomers to the histamine H1 receptor revealed by receptor-bound ligand analysis and molecular dynamics study |
| Journal | : | Journal of Molecular Recognition |
| DOI | : | 10.1002/jmr.3098 |
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society," TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
■
Tokyo University of Science(About TUS)

About Professor Mitsunori Shiroishi
from Tokyo University of Science
Dr. Mitsunori Shiroishi graduated from the Department of Biomolecular Engineering at Tohoku University, Japan, in 1998 and completed his doctoral degree there in 2003. He joined Tokyo University of Science in April 2018, where he currently serves as full Professor. He leads the Protein Engineering Laboratory as Principal Investigator, where his team conducts cutting-edge research on protein science focused mainly on cell receptors with pharmaceutical potential, including receptors with immunoglobin-like fold and G protein-coupled receptors. He has published over 50 research papers on these topics.
Laboratory website 
Official TUS website 
Funding information
This work was funded in part by JSPS KAKENHI (Grant Numbers JP25709080 and JP15K14460) and the Platform Project for Supporting in Drug Discovery and Life Science Research (Platform for Drug Discovery, Informatics, and Structural Life Science) from MEXT and AMED.

