2023.07.06 Thursday
Sweat it Out: Novel Wearable Biosensor for Monitoring Sweat Electrolytes for Use in Healthcare and Sports
The device transmits measurements wirelessly and can be seamlessly applied to the outer surface of textiles
Wearable sensors are becoming a promising tool in personalized healthcare and exercise monitoring. In a recent study, researchers from Japan develop a novel wearable chemical sensor capable of measuring the concentration of chloride ions in sweat. By using a heat-transfer printing technique, the proposed sensor can be applied to the outer surface of common textiles to prevent skin irritation and allergies, and could also be useful in the early detection of heat stroke and dehydration.
The remarkable level of miniaturization possible in modern electronics has paved the way for realizing healthcare devices previously confined to the realm of science fiction. Wearable sensors are a prominent example of this. As the name suggests, these devices are worn on the body, usually directly on the skin. They can monitor important bodily parameters, including heart rate, blood pressure, and muscle activity.
Some wearable sensors can also detect chemicals in bodily fluids. For instance, sweat biosensors can measure the concentration of ions in sweat, providing information on their levels in blood. However, designing such chemical sensors is more complex than physical sensors. Direct contact between a wearable chemical sensor and skin can cause irritation and allergies. In contrast, if the sensor is fabricated directly on a wearable textile, its accuracy decreases due to surface irregularities.
In a recent study, a research team, led by Associate Professor Isao Shitanda of the Tokyo University of Science (TUS) in Japan, has developed an innovative sweat biosensor that addresses the aforementioned problems. Their work,published online in ACS Sensors on June 15, 2023, describes the use of a technique called "heat-transfer printing" to fix a thin, flexible chloride ion sensor onto a textile substrate. The study was co-authored by Dr. Masahiro Motosuke, Dr. Tatsunori Suzuki, Dr. Shinya Yanagita, and Dr. Takahiro Mukaimoto of TUS.
"The proposed sensor can be transferred to fiber substrates, and thus can be incorporated into textiles such as T-shirts, wristbands, and insoles," explains Dr. Shitanda. "Further, health indicators such as chloride ion concentration in sweat can be measured by simply wearing them."
The heat-transfer printing approach offers several advantages. For one thing, the sensor is transferred outside of the piece of clothing, which prevents skin irritation. In addition, the wicking effect of the textile helps spread the sweat evenly between the electrodes of the sensor, creating a stable electrical contact. Moreover, printing the sensor on a flat surface and then transferring it prevents the formation of blurred edges that commonly occur when printing directly onto a textile.
The researchers carefully selected the materials and electrochemical mechanisms of the sensor to avoid risking an allergic reaction for the wearer. After developing the sensor, they conducted various experiments using artificial sweat to verify its accuracy in measuring chloride ion concentration. The change in the electromotive force of the sensor was −59.5 mTV/log CCl−. Additionally, it displayed a Nernst response and a linear relationship with the concentration range of chloride ions in human sweat. Moreover, no other ions or substances typically present in sweat were found to interfere with the measurements.
Lastly, the team tested the sensor on a volunteer who exercised on a static bicycle for 30 minutes, by measuring their perspiration rate, chloride ion levels in blood, and saliva osmolality every five minutes to compare with the data previously gathered by the sensor. The proposed wearable sensor could reliably measure the concentration of chloride ions in sweat.
The sensor can also transmit data wirelessly, making it useful for real-time health monitoring. "Since chloride is the most abundant electrolyte in human sweat, measuring its concentration provides an excellent indicator of the body's electrolyte balance and a useful tool for the diagnosis and prevention of heat stroke," remarks Dr. Shitanda.
This research thus demonstrates the potential of using wearable ion sensors for the real-time monitoring of sweat biomarkers, facilitating personalized healthcare development and athlete training management.
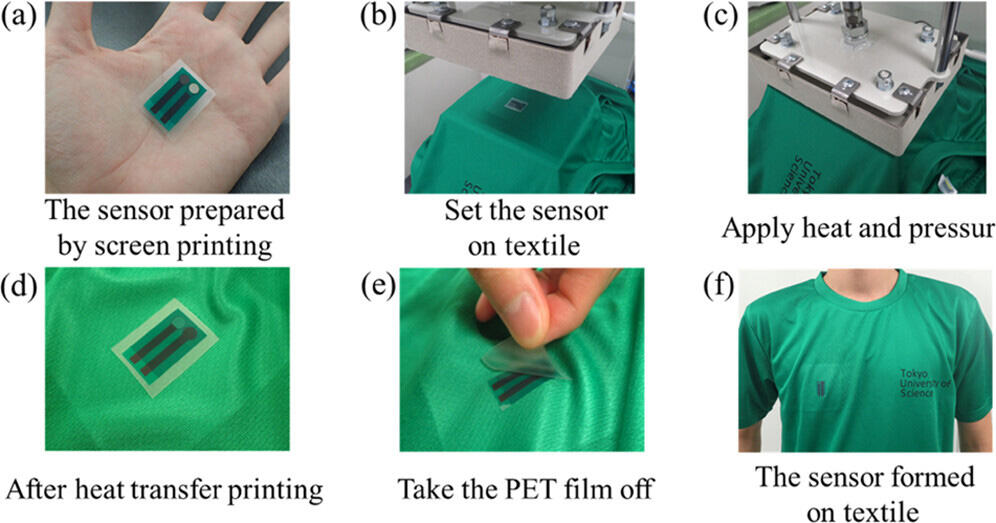
Image Title: Heat-transfer printing process for fixing the chloride ion sensor onto a textile substrate.
Image Caption: The proposed wearable sensor for measuring chloride ion levels in sweat can be easily applied to textiles using the heat-transfer printing technique, preventing skin irritation and allergies.
Image Credit: Isao Shitanda from TUS, Japan
License type: CC BY 4.0
Reference
| Title of original paper | : | Wearable Ion Sensors for the Detection of Sweat Ions Fabricated by Heat-Transfer Printing |
| Journal | : | ACS Sensors |
| DOI | : | 10.1021/acssensors.3c01027 |
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society," TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
■
Tokyo University of Science(About TUS)

About Associate Professor Isao Shitanda from Tokyo University of Science
Isao Shitanda graduated from the Tokyo University of Science (TUS) in 2001 and received a Ph.D. from The University of Tokyo in Japan in 2006. Since 2012, he has been an Associate Professor at the Department of Pure and Applied Chemistry at TUS, where he leads the Itagaki/Shitanda lab. He specializes in electrochemical micro/nano systems, physical and analytical chemistry, bio-related chemistry, and environmental chemistry, among other fields. He has published over 180 papers and holds six patents.
Laboratory website

Official TUS website

Funding information
This study was partially supported by JST-ASTEP Grant Number JPMJTS1513, JSPS Grant Number 17H02162, the Private University Research Branding Project (2017−2019) from the Ministry of Education, Culture, Sports, Science and Technology, and the Tokyo University of Science Grant for President's Research Promotion.

