2022.04.21 Thursday
Bonds From the Past: A Journey Through the History of Protein Synthesis
Scientists provide evolutionary insights into protein synthesis using primordial transfer RNA and ribosomes
The process of "translation" in protein synthesis involves formation of a peptide bond between two amino acids that are attached to two distinct transfer RNAs (tRNAs). For long, scientists have been puzzled as to how these tRNAs evolutionarily lie so close to each other on the ribosome. In a new study, researchers explain how tRNA-like components act as scaffolds for peptide bond formation between amino acid-bound "RNA minihelices," which are half tRNA-like molecules.
The genetic information stored in DNA is "decoded" to form proteins via the process of translation. This involves the formation of peptide bonds between amino acids bound to transfer RNA (tRNA) molecules that glide over the ribosome in very close proximity to each other, and elongate the peptide chain, which later undergoes conformational change, forming a protein. In contrast to the codon-dependent aminoacyl-tRNA recognition in the small ribosomal subunit, the peptide bond formation in question occurs at the peptidyl transferase center (PTC) of the large ribosomal subunit, in a non-amino acid specific manner. This non-specificity indicates that the large subunit evolved before the small subunit, which has more specific interactions with mRNA and tRNA.
Although the evolutionary process of PTC formation has been thoroughly documented, little is known about how ribosomes developed into functioning entities and became an essential component of protein synthesis. Scientists have long been perplexed by the fact that tRNAs require the help of a "scaffold" in order to create a peptide bond, which orients them for interaction via 3'-CCA sequences on their acceptor arms. What that scaffold is, and how it operates, would be intriguing to learn about.
A team of scientists at Tokyo University of Science, led by Prof. Koji Tamura, decided to solve this mystery using a perspective of continuity in biological evolution. Their study, which was published online on 12 April 2022 in Volume 12, Issue 4 of the journal Life, sheds light on the evolutionary aspect of protein translation. Their results represent important evidence to prove the hypothesis about the origin and evolution of the PTC, which has changed the way we look at the modern-day ribosomes and tRNA.
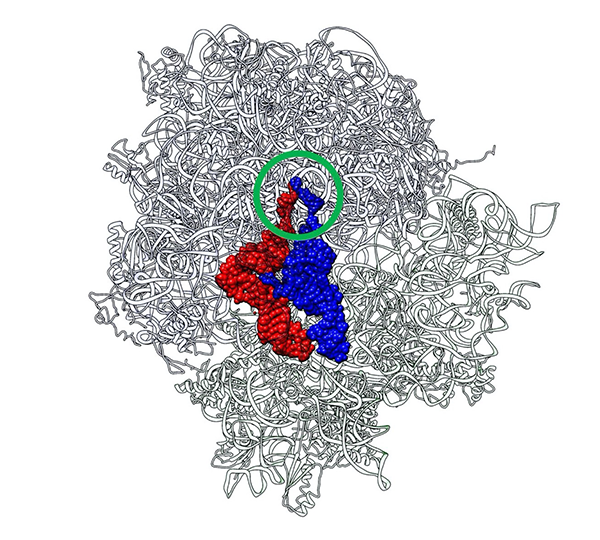
The idea sprang to life after taking a close look at the crystal structure of the 70S ribosome-tRNA complex from Thermus thermophilus, a bacterium often used in the study of genetics. The peptidyl (P-) and aminoacyl (A-) sites of the tRNAs here aligned to bring the CCA termini in close proximity, like a rugby player's index fingers in the "Goromaru pose." "There was a certain entity that served as a scaffold for maintaining this proximity, and it most likely stemmed from the primordial PTC," says Prof. Tamura. Since an evolutionary aspect was likely, the team chose to utilize primordial tRNA or "RNA minihelix" for their study.
|
Title: The structure of Thermus thermophilus' 70S ribosome in complex with tRNAs
Caption: Two CCA termini (circled in green) of P-site tRNA (red) and A-site tRNA (blue) approach in close proximity just like a "Goromaru pose". The figure was drawn using a coordinate file with PDB ID: 4V5D.
Image Credit: Koji Tamura from Tokyo University of Science
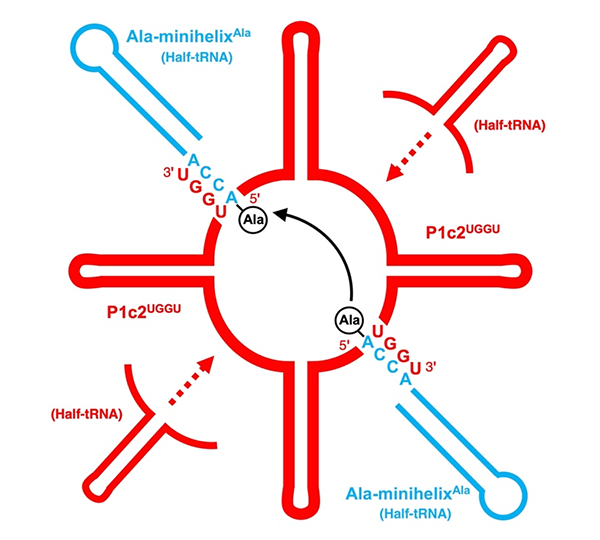
They first attempted a peptide bond formation between two alanine-specific minihelices in the presence of a ribosomal RNA segment. The peptide bond was formed using the ribosomal segment, P1c2, as an RNA scaffold which was just 70 nucleotides long! Next, they added a terminal amino acid segment (with the sequence UGGU) to the P1c2 (P1c2UGGU). According to mass spectrometry results, this increased the peptide bond formation ability by 4.2 times that of the original! The peptide bond formation between two alanine residues was supported by a scaffold of dimerized P1c2UGGU. The UGGU sequence of the scaffold interacted with the corresponding 3'-terminal ACCA of the minihelix and brought the two amino acids near enough to create a peptide bond. Nobel laureate Dr. Ada Yonath and her group recently showed that similar, conserved PTC regions could catalyze peptide bond formation with artificial analog molecules, but Prof. Tamura's group showed that an aminoacylated RNA could also be a substrate.
The findings definitely imply a possibility that minihelices bind to the primordial PTC. So, what do the results suggest about the evolution of ribosomes? "Functional interactions between the CCA of tRNA and PTC could have been 'revised' in the process of evolution. Although current ribosomes do not have a contiguous sequence like UGGU, their interactions are 'conceptually' similar to the effects seen in our study. It is plausible that minihelices eventually evolved into tRNA using, for example, kissing-loop interactions between two minihelix-like RNA molecules," Prof. Tamura explains. "These minihelix-like molecules, which form a part of the scaffold for peptide bond formation, may have not only contributed to the evolution of what is currently the PTC, but also formed tRNA molecules," he adds.
The future applications of this research―which has opened up exciting avenues in evolutionary RNA biology―are manifold. Faced with a metabolic paradox (that the components of DNA and RNA are generated from amino acids), it is conceivable to investigate the notion of "peptide nucleic acids" as genetic material precursors. The results are fascinating, and they will help scientists to decode molecular phenomena that have eluded them for years.
|
Title: Representation of peptide bond formation between two minihelices
Caption: This schematic diagram explains how two alanyl-minihelicesAla, use dimerized P1c2UGGU, as a scaffold for peptide bond formation.
Image Credit: Koji Tamura from Tokyo University of Science, with permission from MDPI
Reference
| Title of original paper | : | Peptide bond formation between aminoacyl-minihelices by a scaffold derived from the peptidyl transferase center |
| Journal | : | Life |
| DOI | : | 10.3390/life12040573 |
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society", TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
About Professor Koji Tamura from Tokyo University of Science
Dr. Koji Tamura is a Professor at the Department of Biological Science and Technology at the Tokyo University of Science with 16 years of teaching experience. He received his Ph.D. from the University of Tokyo and did his postdoctoral work at The Scripps Research Institute with Professor Paul Schimmel. His research involves multidisciplinary study of the origin of life on Earth. His area of interest lies in the subjects like analyses of aminoacyl-tRNA synthetases mediated aminoacylation of tRNA, its relationship to the origin of the genetic code, and the origin of peptide bond formation on the ribosome.
https://www.tus.ac.jp/en/fac/p/index.php?4d37 
https://kojitamura.web.fc2.com/ 
Funding information
This study was supported by the Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS) (Grant No. 17K19210 and 21K06293 to K.T., and Grant No. 19K16204 to H.M.-A.).

