2021.08.10 Tuesday
Striking Gold: A Pathway to Stable, High-Activity Catalysts from Gold Nanoclusters
Researchers design a stable, high-performance photocatalyst from gold nanoclusters by removing the protective molecules around them
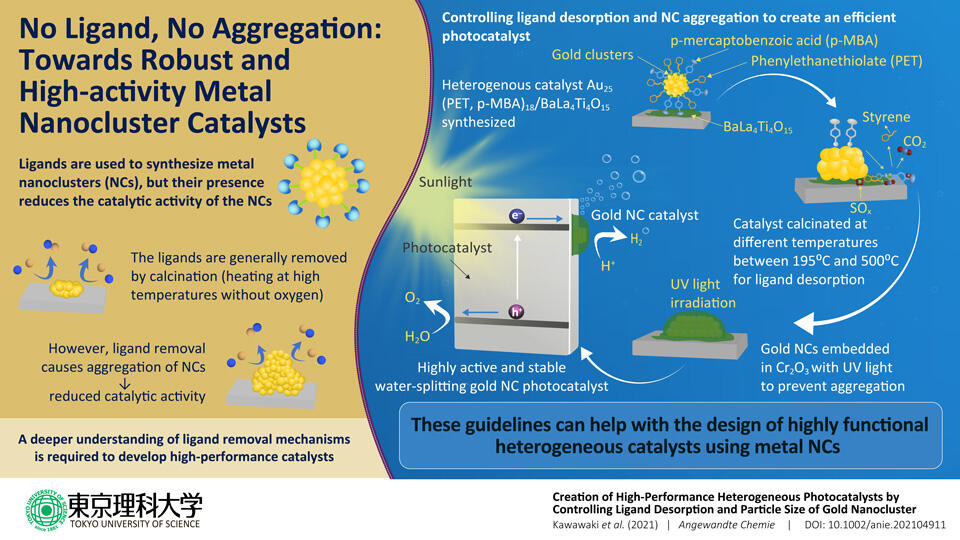
Precise metal nanoclusters (NCs) are ideal for developing practical catalysts for chemical reactions. However, their catalytic activity is reduced either due to protective molecules called "ligands" surrounding them or aggregation resulting from ligand removal. In a new study, scientists from Japan elucidate the ligand removal mechanism for gold NCs and irradiate them with UV light to prevent aggregation, creating a high-functioning photocatalyst.
|
Catalysts, which help drive reactions, are ubiquitous, whether as an enzyme in the body that digests food or the catalytic converter in the car that breaks down pollutants. In chemical reactions, catalysts play an important role in making it more efficient. Recently, atomically precise metal nanoclusters (NCs) that can accelerate various thermal, electrochemical, and photochemical reactions have been used to design useful catalysts. These NCs are tiny particles (less than 2 nanometers) whose properties can be modified by changing their atomic composition. This is why, metal NCs have received considerable attention, with scientists trying to find various ways of synthesizing NCs with unique functions.
A popular way of fabricating atomically precise metal NCs is using ligands (molecules or ions that attach themselves to a central metal core). These ligands not only protect the tiny NCs but also affect their chemical reactivity and selectivity. Sometimes, however, the reactivity is lower than expected.
To increase the catalytic activity of ligand-protected metal NCs, they are heated in a furnace at high temperatures without oxygen (a process called "calcination") to remove the ligands from the main cluster. However, heating the particles at very high temperatures can cause the NCs to accumulate, often leading to a decrease in reactivity. "When the ligands are removed without special treatment, the metal NCs easily aggregate on the support and lose their size-specific properties. It is essential understand the mechanism of ligand calcination to create highly functional heterogeneous catalysts under appropriate conditions," says Prof. Yuichi Negishi of Tokyo University of Science, Japan, who researches on the synthesis of nanoclusters.
In a new study published in Angewandte Chemie, Prof. Negishi led a team of researchers, including Assistant Professor Tokuhisa Kawawaki, Mr. Yuki Kataoka, Ms. Momoko Hirata, and Mr. Yuki Akinaga, to dig deep into the mechanism of the ligand removal process in NCs. For their experiments, the researchers synthesized gold NCs protected by two ligands, 2-phenylethanethiolate and mercaptobenzoic acid and then supported them on a photocatalytic metal oxide. Next, the team heated the prepared material at different temperatures ranging from 195°C to 500°C. After every step, they analyzed the products using techniques such as infrared spectroscopy, x-ray photoelectron spectroscopy, and transmission electron microscopy to identify the changes in their chemical composition.
After the ligands were completely released, the team embedded the gold NCs within a thin film of chromium oxide by irradiating the sample with UV light in order to prevent aggregation of the NCs. This process generated a photocatalyst with useful properties like high water-splitting activity and stability.
These findings guide the design for metal NC-based catalysts in the future, with applications in hydrogen generation for hydrogen fuel cells. "With our research, we hope to build a clean, sustainable, society, one brick at a time," concludes Prof. Negishi.
It may not be too far into the future before his vision is realized!
Reference
| Title of original paper | : | Creation of High-Performance Heterogeneous Photocatalysts by Controlling Ligand Desorption and Particle Size of Gold Nanocluster |
| Journal | : | Angewandte Chemie |
| DOI | : | 10.1002/anie.202104911 |
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society", TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
About Professor Yuichi Negishi from Tokyo University of Science
Dr. Yuichi Negishi is a Professor in the Department of Applied Chemistry at Tokyo University of Science. As senior researcher, he has more than 190 publications to his credit (total citations are over 12,000 times) and is the head of his research laboratory at the university. His areas of research include physical chemistry, cluster chemistry, and nanomaterial chemistry. His notable achievements include The Chemical Society of Japan Award for Young Chemists (Japan Chemical Society, 2008), the Japan Society for Molecular Science Award for Young Scientists (Japan Society for Molecular Science, 2012), Yagami Prize (Keio University, 2017), Distinguished Award 2018 for Novel Materials and Their Synthesis (IUPAC etc., 2018) and International Investigator Awards of the Japan Society for Molecular Science (Japan Society for Molecular Science, 2020)
https://www.tus.ac.jp/en/fac/p/index.php?5825 
https://www.rs.kagu.tus.ac.jp/negishi/ 
About Assistant Professor Kawawaki Tokuhisa from Tokyo University of Science
Funding information
This study was funded by the Japan Society for the Promotion of Science (JSPS; grant numbers JP16H04099, 16K21402, 20H02698, and 20H02552).

