2020.01.15 Wednesday
New study on a recently discovered chlorophyll molecule could be key to better solar cells
Scientists uncover the location and functions of a new type of chlorophyll molecule for the first time
 |
Photosynthesis, the process by which some organisms convert sunlight into chemical energy, is well known. But, it is a complex phenomenon, which involves a myriad of proteins. The molecule Chlf, a new type of chlorophyll, is known to play a part in photosynthesis, but owing to its recent discovery, its location and functions are not understood. Scientists from Japan have now analyzed in detail the protein complex involved in photosynthesis and uncovered several new aspects about Chlf.
All living organisms need energy for their survival, and this energy indirectly comes from the sun. Some organisms, such as plants, cyanobacteria, and algae, are capable of directly converting this light energy into chemical energy via a process called "photosynthesis". These photosynthetic organisms contain special structures to mediate photosynthesis, called "photosystems". There are two photosystems that carry out light-energy conversion reactions, each of which is composed of a number of proteins and pigments. Among photosynthetic pigments, chlorophyll is the most crucial one, which not only captures light energy from the sun but also participates in the "electron transfer chain", a molecular pathway through which photons (from the sunlight) are converted into electrons (which are used as an energy source). There are different types of chlorophyll molecules, each having a specific function ranging from absorbing light and converting it into energy. Moreover, each chlorophyll molecule absorbs light in different regions. Recently, a new type of chlorophyll called Chlf was discovered, but details like exactly where it is located and how it functions have remained a mystery until now.
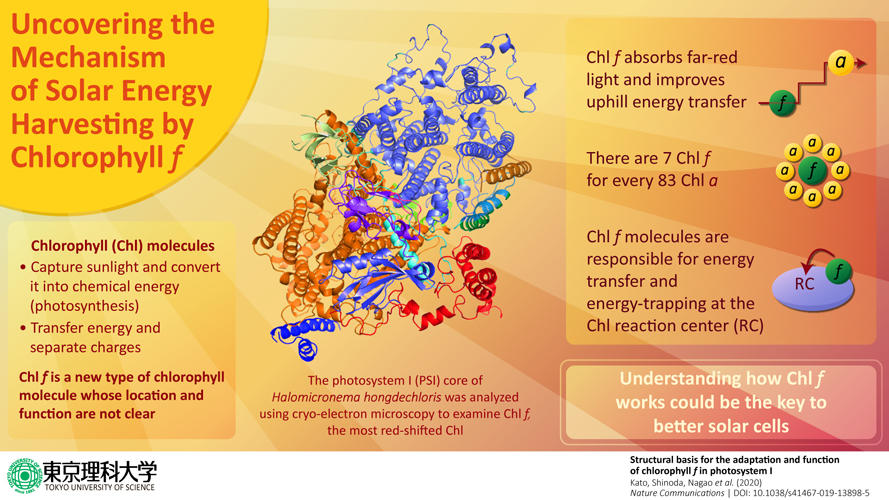
In a new study published in Nature Communications, a team of researchers led by Prof. Tatsuya Tomo at the Tokyo University of Science, Japan, and including collaborating researchers from Okayama University, Tsukuba University, Kobe University, and RIKEN, revealed new details about the location and functions of Chlf. They wanted to gain insight into the complex process of photosynthesis, as an in-depth understanding of this process could have various future applications, such as the development of solar cells. Talking about the study, Prof. Tomo says, "The initial course of photosynthesis begins when the photosynthetic pigment bound to this photochemical complex absorbs light. We analyzed the structure of a newly discovered photochemical complex, photosystem I with Chlf that has an absorption maximum on the lower energy side of light (far-red light). Moreover, we analyzed the function of Chlf."
What the scientists knew so far was that Chlf is "far-red shifted," which means that this molecule absorbs far-red light from the lower end of the light spectrum. Prof. Tomo and his team wanted to dig deeper, and for this, they studied the alga in which Chlf was first discovered. By using techniques such as cryo-electron microscopy, they analyzed the high-resolution structure of the photosystem in this alga in detail and found that Chlf is located at the periphery of photosystem I (one of the two types of photosystems) but is not present in the electron transfer chain. They also found that far-red light causes structural changes in the photosystem, which are accompanied by the synthesis of Chlf in the algae, leading them to conclude that Chlf causes these structural changes in photosystem I. This was exciting, as this finding is the first to explain how exactly Chlf works. Prof Tomo says, "Our findings revealed that the appearance of Chlf is well correlated with the expression of photosystem I genes induced under far-red light. This indicates that Chlf functions to harvest the far-red light and enhance up-hill energy transfer. We also found that the amino acid sequence of photosystem I was altered so as to accommodate the structure of Chlf."
Understanding the intricacies of photosynthesis has several important applications. For example, mimicking the process of photosynthesis in an artificial system is an elegant method of capturing solar energy and converting it into electricity. Prof Tomo elaborates, "About half of the solar energy that falls on the earth is visible light, and the other half is infrared light. Our research puts forth a mechanism that can use light on the lower energy spectrum, which has never been seen before. Our findings show how to improve the efficiency of energy transfer in photosynthesis and, by extension, also provide important insights into artificial photosynthesis."
Reference
| Authors | : | Koji Kato1,14, Toshiyuki Shinoda2,14, Ryo Nagao1,14, Seiji Akimoto3, Takehiro Suzuki4, Naoshi Dohmae4, Min Chen5, Suleyman I. Allakhverdiev6,7,8,9,10, Jian-Ren Shen1*, Fusamichi Akita1,11*, Naoyuki Miyazaki12,13*, and Tatsuya Tomo2* |
| Titles of original paper | : | Structural basis for the adaptation and function of chlorophyll f in photosystem I |
| Journal | : | Nature Communications |
| DOI | : | https://www.nature.com/articles/s41467-019-13898-5 |
| Affiliations | : |
1Research Institute for Interdisciplinary Science and Graduate School of Natural Science and Technology, Okayama University, Japan 2Faculty of Science, Tokyo University of Science, Japan 3Graduate School of Science, Kobe University, Japan 4Biomolecular Characterization Unit, RIKEN Center for Sustainable Resource Science, Japan 5School of Life and Environmental Sciences, University of Sydney, Australia 6К.А. Timiryazev Institute of Plant Physiology RAS, Russia 7Institute of Basic Biological Problems RAS, Pushchino, Russia 8M.V. Lomonosov Moscow State University, Russia 9Moscow Institute of Physics and Technology, Russia 10Institute of Molecular Biology and Biotechnology ANAS, Azerbaijan 11Japan Science and Technology Agency, PRESTO, Japan 12Institute for Protein Research, Laboratory of Protein Synthesis and Expression, Osaka University, Japan 13Life Science Center for Survival Dynamics, Tsukuba Advanced Research Alliance, University of Tsukuba, Japan 14These authors contributed equally: Koji Kato, Toshiyuki Shinoda, Ryo Nagao |
*Corresponding author's email: tomo[@]rs.tus.ac.jp; shen[@]okayama-u.ac.jp; fusamichi_a[@]okayama-u.ac.jp; naomiyazaki[@]tara.tsukuba.ac.jp;
※Please change [@] to @.
Media contacts
Okayama University
Email: www-adm[@]adm.okayama-u.ac.jp
Website:http://www.okayama-u.ac.jp/index_e.html
University of Tsukuba
Email: kohositu[@]un.tsukuba.ac.jp
RIKEN
Email: ex-press[@]riken.jp
Kobe University General Affairs Department Communications Division
E-mail: ppr-kouhoushitsu[@]office.kobe-u.ac.jp
※Please change [@] to @.
About The Tokyo University of Science

Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan's development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of "Creating science and technology for the harmonious development of nature, human beings, and society", TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today's most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
Funding information
This study was supported by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) from AMED, PRESTO from JST Grant no. JPMJPR16P1, JSPS KAKENHI no. JP17K07442, JP19H04726, JP17H06434, no. 17726220801, 17K07453, 18H05177, JP19K22396, along with the JSPS-RFBR grant (no. 19-54-50002).
PROFILE
About Professor TOMO Tatsuya
Tokyo University of Science, Faculty of Science Division I, Liberal Arts:
https://www.tus.ac.jp/en/fac/p/index.php?5d5b
https://www.rs.tus.ac.jp/tomo/english.html

